Duellen: Eli Lilly slår Novo Nordisk
Ända sedan Eli Lilly släppte resultaten från sina tester – där patienter tappade mer än 20 procent av sin kroppsvikt – har investerare fått upp ögonen för läkemedlet Mounjaro. Även Novo Nordisks dito Ozempic väcker stort intresse. Marknaden för viktminskning är stor och det är liten tvekan om att de nya läkemedlen kommer bli de bäst säljande på ett decennium, skriver Barron’s.
Men för investerare är det dags att sålla ut investeringsmöjligheterna i hajpen, skriver tidningen och ställer aktierna mot varandra. Köp Eli Lilly, lyder rådet efter duellen.
Weight-Loss Drugs Will Be Blockbusters. Here’s the Stock to Buy.
Everyone is talking about Eli Lilly’s Mounjaro and Novo Nordisk’s Ozempic. Now, the drugs are poised to go from conversation starters to profit makers.
In the year since Eli Lilly announced trial results showing that tirzepatide—best known by the brand name Mounjaro—helped patients drop more than 20% of their weight, the drug has become the most talked about, and most hyped, pharmaceutical in recent memory.
In recent months, Lilly’s (ticker: LLY) drug, and a similar one from Novo Nordisk (NVO), have joined the pantheon of culture-reshaping pharmaceuticals that includes the birth control pill, antidepressant Prozac, and erectile dysfunction drug Viagra. Lilly and Novo shares, meanwhile, have soared. Each company now has a market value of around $400 billion, making them the second- and third-largest in all of Big Pharma. That’s a neat trick given that their revenue is about 25% to 30% of that of industry leader Johnson & Johnson (JNJ), which has a market cap of $425 billion.
There’s little doubt that Lilly’s tirzepatide and its Novo competitor, semaglutide, will be some of the best-selling drugs of the decade. For investors, however, it’s time to separate the opportunity from the hype.
Big risks remain for the two companies, including questions of what health benefits they can prove for the medicines, who will pay for them, and what competitors might emerge. And for anyone looking at buying into the companies now, there’s the matter of whether the sky-high expectations for the drugs are already reflected in the companies’ share prices, or if there’s still room for upside.
Below, Barron’s guide to navigating this new era of obesity medicine.
What’s so revolutionary about these drugs?
The first thing to know is that we’re talking about two drugs—Lilly’s tirzepatide and Novo’s semaglutide—which are marketed under a variety of names and formulations, depending on the condition they are intended to treat.
Novo sells semaglutide as Ozempic, an injection for Type 2 diabetes; Wegovy, an injection for obesity; and Rybelsus, a pill for Type 2 diabetes.
For now, Lilly’s tirzepatide is sold only as Mounjaro, an injection to treat Type 2 diabetes. But the company will likely have a new name to add to its marketing roster soon: In late April, it announced results of a Phase 3 trial finding that Type 2 diabetes patients using the drug lost 15.7% of their body weight when taking tirzepatide. Lilly says it will finalize its application for approval of the drug as an obesity treatment and, if all goes smoothly, the FDA nod could come as soon as the end of this year.
Both drugs are significantly better at lowering blood sugar and promoting weight loss than previous medicines. They work by mimicking natural hormones that trigger the body to produce more insulin and slow digestion, making people feel fuller for longer.
Weight loss, in particular, has been a struggle for drugmakers. Virtually all previous attempts have been ineffective and/or dangerous; the list of weight loss drugs pulled from the market over safety concerns is a long one.
But Novo and Lilly’s drugs appear to be safe (though both have significant side effects) and their efficacy is remarkable. Patients on Lilly’s tirzepatide lost 22.5% of their body weight on average in one trial, while patients on the Novo drug lost 14.9% of their body weight in a separate trial. By comparison, an older Novo drug called Saxenda, which went on the market about a decade ago, cut patients’ body weights by only 7.4%.
When will the drugs be available?
Lilly’s Mounjaro and Novo’s Ozempic, Wegovy, and Rybelsus are already on the market, though high demand has kept them in short supply. Both companies have said they are ramping up production. Novo said in April it had a new contract manufacturer ready to go, and Lilly has said it would double its capacity to make Mounjaro and similar drugs by the end of the year.
If Lilly’s bid for approval to use tirzepatide for weight loss is approved by the FDA, the drug would likely be available immediately. (Meanwhile, there are widespread reports that Mounjaro is already being used for that purpose off-label.)
Are competitors coming?
Probably. Pfizer (PFE) says it has a pill under development that works similarly to tirzepatide and semaglutide, while Amgen (AMGN) is testing a weight-loss drug that uses a different mechanism. Lilly also has a number of other weight-loss drugs in its pipeline, including a pill called orforglipron, which could launch in 2027; SVB Securities analyst David Risinger expects it could reach $9.9 billion in sales in 2030.
That said, drug development is an uncertain business. A reminder of that fact came this March, when shares of the biotech Altimmune (ALT) plunged after the company disclosed that a significant share of patients in a closely watched trial of its new weight-loss drug dropped out due to gastrointestinal issues.
Regardless, analysts say the weight-loss market is big enough to accommodate a number of drugs. In a note last year, Cowen analyst Michael Nedelcovych wrote that he expects obesity drug sales to hit $30 billion by 2030—a number that doesn’t include the Type 2 diabetes indication. “The market is going to be very significant, if indeed these companies can prove that there are substantial health benefits beyond just the weight loss,” says Risinger.
Just how big could the overall market be?
At an investment conference early this year, Pfizer CEO Albert Bourla said that his company expects the combined obesity and Type 2 diabetes market for drugs in this category to hit $90 billion globally by 2030. That kind of estimate is supported by the extremely widespread nature of the two conditions: According to the Centers for Disease Control and Prevention, 41.9% of American adults are obese, and about 1 in 10 have diabetes.
And because these medicines aren’t curative, patients will probably need to take them for a long time, expanding the drugs’ earnings potential. For example, analyst expectations for peak sales of Mounjaro—in both its diabetes and obesity formulations—include a heady $100 billion a year (Geoff Meacham of BofA Securities) and a more modest, but still impressive, $40 billion (Jefferies analyst Akash Tewari). For context: Even the smaller of those figures is nearly twice peak sales of the best-selling therapeutic in history, AbbVie’s (ABBV) Humira.
How much do these drugs cost, and who will pay?
The drugs cost around $1,000 a month, depending on indication and dosing. For patients prescribed the drugs to treat diabetes, payment works as it does for most medicines: Drugmakers negotiate insurance coverage with pharmacy benefit managers, and coverage builds slowly over time. Today, just under 60% of people on commercial insurance plans and Medicare have access to Mounjaro, according to Lilly.
When the drugs are prescribed as obesity treatments, though, it’s a different story. Lingering debates over whether obesity should be considered a disease mean insurers often don’t cover weight-loss drugs, and federal law bars Medicare’s prescription drug benefit from paying for them.
Investors and analysts say they expect insurer coverage to expand in the face of patient demand. Still, private insurers have signaled that even as they begin to cover Novo’s drug for obesity (the company says around 40 million Americans now have access to Wegovy through their insurance), not every overweight person will be eligible. “We need to really be clear about which patients really do benefit from these medicines, and make sure we properly understand how they’re going to use those medicines,” Andrew Witty, CEO of UnitedHealth Group (UNH), one of the country’s largest health insurers, said on an investor call in April. Days later, an executive at health insurer Elevance Health (ELV) said the company doesn’t cover weight-loss drugs, except when required to by state law.

Lilly and Novo haven’t yet gotten study results proving that their obesity drugs not only help non-diabetics lose weight, but also lower their risk of heart disease and stroke. Both are currently conducting trials to provide this data (Novo’s is expected to produce results in the coming months; Lilly’s is still years out). If the results are positive, it will be much harder for insurers to resist covering the drugs as obesity treatments.
Meanwhile, legislative efforts to roll back the law that stops Medicare from covering weight-loss medications have been ongoing. If they are successful, the program could be in for an enormous bill. A New England Journal of Medicine paper published in March found that if only 10% of eligible Medicare patients took Novo’s drug, it would cost Medicare $26.8 billion a year, equivalent to 18.5% of spending on Medicare’s Part D prescription drug benefit in 2019.
What could go wrong for Novo and Lilly?
The biggest risk for Novo and Lilly is that trials designed to prove that the drugs lower rates of stroke and heart attack in patients without diabetes fail. In that case, insurance coverage in the obesity indications would remain limited. Yes, the drugs would still be megablockbusters, but not on the scale many are expecting.
There are other, less obvious threats. One is the impact of Medicare price negotiations, set to go into effect in 2026 for Medicare’s prescription drug program under last year’s Inflation Reduction Act. Medicare currently covers both Novo and Lilly’s drugs for diabetes, and Novo’s Ozempic could be eligible for negotiation as early as 2027. No one really knows how the negotiations will play out, but it’s possible that lower Medicare prices for Ozempic could also impact the prices Lilly can fetch for its competitive drug.
(Lilly executives note, however, that a head-to-head trial comparing weight loss in non-diabetic obese and overweight patients on Novo’s drug and Lilly’s drug is slated to produce data by 2025. If Lilly were to win that face-off, it could help the company maintain higher prices.)
Then there are the threats from competitors. If the forthcoming Pfizer or Amgen drug were to match or outperform Novo and Lilly’s products, those glowing analyst projections might come down to earth.
Are Lilly and Novo shares a good bet?
Bulls see an enormous long-term opportunity in Lilly. “Can Mounjaro, plus the pipeline of products that Lilly is going to launch into the obesity market over the next five to 10 years, create a massive sales opportunity that is here to stay?” asks Debra Netschert, a managing director at Jennison Associates, who is one of the managers of the PGIM Jennison Health Sciences fund (PHLAX). “I believe it can.”
Lilly shares doubled from the start of 2021 through the end of 2022, and the stock has continued to climb. This week saw Lilly shares hit an all-time high as the company reported positive results on a Phase 3 trial of its Alzheimer’s disease therapy. Analysts are mixed on the full implications of the new data, and there are real safety issues with the drug, but Lilly avoided a worst-case scenario of the study failing, removing what had been a near-term worry for investors.
Novo shares have also more than doubled over the past two years, and the company says its Wegovy rollout is picking up steam as supply capacity increases.
Heavyweights
A new class of obesity ans diabetes drugs have helped Lily and Novo shares outperform.
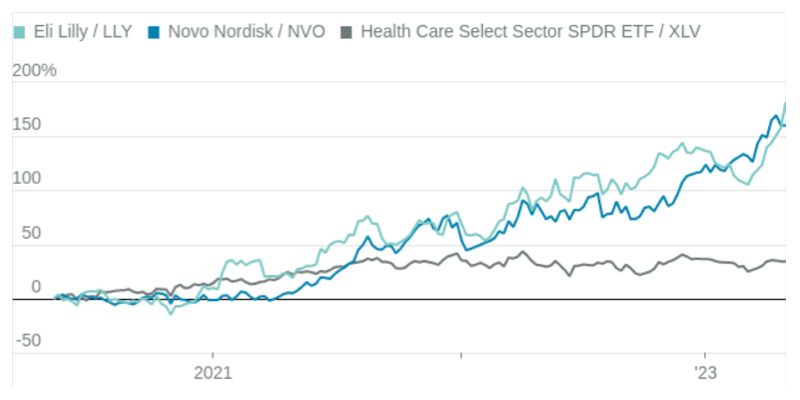
For investors looking to make a bet on the obesity drugs, Lilly is the more promising of the two companies, despite the stock run-up. Analysts predict Lilly’s earnings per share to grow by 23% on a compound annual basis from 2022 through 2027, according to FactSet—better than Novo’s 17%, and well above expectations for the rest of the sector. Merck, by contrast, is expected to grow earnings by 8% over the same period.
“We’re talking about a company that has extraordinary growth potential, probably more than four times that of the pharmaceutical group over all,” says SVB’s Risinger.
And while both companies have robust drug pipelines, Lilly’s has a slight edge. In addition to its obesity pill and retatritude, another obesity injectable, the company has a host of potentially major drugs in the late stages of development, including the Alzheimer’s drug donanemab, newly-approved cancer drug Jaypirca, and dermatitis treatment lebrikizumab.
Combine those factors with the early data suggesting that Lilly’s tirzepatide is a more effective weight-loss agent than Novo’s Ozempic (a head-to-head trial in the works), and Lilly looks to be a winning bet on the obesity-drug boom.